Manasa Gadde
My research is focused on investigating the role of the tumor microenvironment in tumor development, progression and metastasis. Specifically, I am focusing on inflammatory breast cancer (IBC), a rare and very aggressive type of breast cancer. IBC presents itself as diffuse emboli of tumor clusters collectively migrating throughout the breast tissue and into surrounding vessels and skin rather than as a solid mass. It is characterized by high rates of metastasis and treatment resistance. Currently, there is no treatment specifically for IBC due to a lack of widely accepted histological and molecular markers unique to IBC. There is tremendous need for physiologically representative system of IBC to study key tumor-stromal interactions and microenvironmental triggers in a systematic and high throughput manner for determination of therapeutical IBC targets. My goal is to create, tune, and utilize a first-of-its-kind physiologically representative three-dimensional (3D) in vitro tumor platform for modeling IBC emboli and collective emboli migration as well as their interactions with nearby blood and lymphatic vessels to identify targetable stromal-tumor cell interactions driving the classic IBC phenotype of emboli collective migration. Ultimately this project will elucidate the key role of the breast tissue microenvironment on IBC progression to determine targets for early diagnosis and therapeutic intervention.
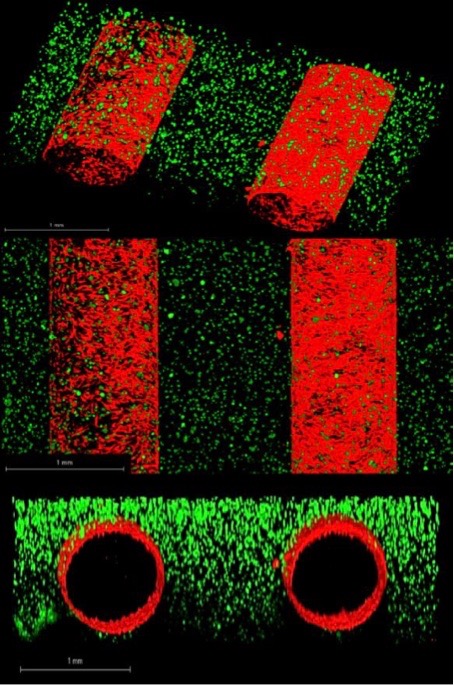
Figure 1: 3D In vitro vascularized dual vessel breast tumor platforms consisting of MDA-IBC3 (green) breast cacner cells in a collagen matrix around two vessels seeded with telomerase immortalized microvascular endothelial (TIME) cells (red). Each panel illustrates different views of the same platforms. Scale bar: 1 mm.