Featured Scientist

Jianchen Yang
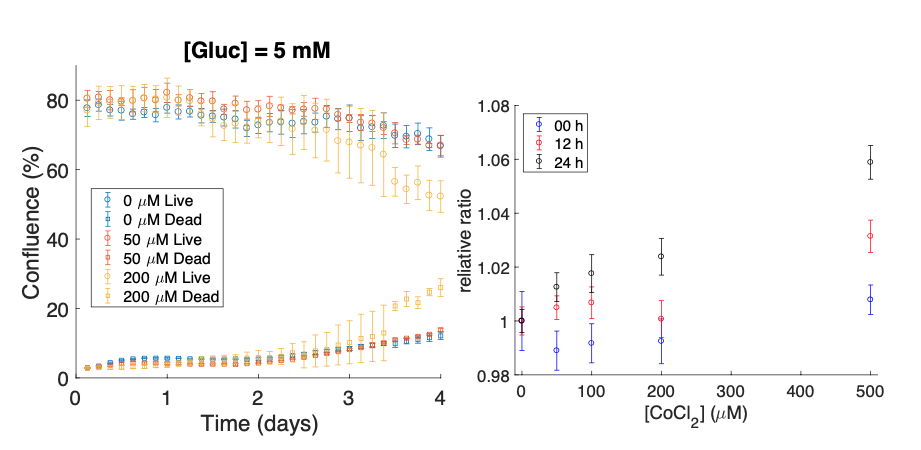
The overall goal of my research is to develop and validate a mathematical model that predicts how the dynamics of some critical nutrients (oxygen, glucose, and lactate) could influence tumor cell metabolism and tumor development. Therefore, we are working to develop a computational-experimental approach to model the proliferation and death of tumor cells in response to environment nutrient dynamics, as well as nutrient variance due to consumption and production. As we cannot adjust the oxygen levels in our current imaging system, I have been developing an experimental model studying the effect of cobalt chloride to induce hypoxia in breast cancer (MDA-MB231) cells in vitro. Using time-resolved microscopy, I have tracked the changes in tumor cell growth when the cells were supplied with different initial glucose and cobalt chloride levels. We found that there is a significant difference between the growth curves of the cells for negative controls and those treated with 200 μM of cobalt chloride. Using MDA-MB231 cells transfected with a lactate reporter, via FRET (fluorescence resonance energy transfer) technology, we also evaluated lactate change. For each cobalt chloride level, the FRET ratio increased over 24 hours, where the highest cobalt chloride level increased the fluorescence ratio the most (500 μM). This suggests that cobalt chloride is inducing hypoxia in the tumor cells, leading to more glycolysis and lactate accumulation.
Cobalt chloride induced hypoxia alters tumor cell metabolism and growth. Left panel: cell confluence over time for live and dead cells for one glucose level and various cobalt chloride levels. Right panel: the relative ratios for the lactate reporter per cobalt chloride dose at three different times.